Since then, a quarter century of rigorous, innovative MIT research on the nature of memory has substantiated that pledge. While huge and tantalizing questions remain, Picower Institute researchers have made major contributions to the field’s understanding of how memory works and how the brain uses memory to survive and thrive. They have mastered the molecular, cellular and circuit mechanisms of memory systems enough to demonstrate groundbreaking feats of mind reading, memory restoration and even memory manipulation in animals. They have revealed profound intersections of memory and behavior, and explained how we consciously use memory.
Once investigated only as a psychological phenomenon of mind, memory is now also studied biologically and physically, said Picower Professor Susumu Tonegawa, who founded the center (CLM) in 1994 and became the Picower Institute’s inaugural director. That level of inquiry provides a mechanistic foundation for better understanding psychiatric and neurological disorders.
“By identifying at the cellular level where information is stored, the next stage is that we can use the function of these cells to overcome deficiencies,” Tonegawa said.
The new depth of insight has also allowed neuroscientists to study how the brain employs memory in intelligent behavior, said Matt Wilson, associate director of the Picower Institute. “The primary function of memory in biological systems is to understand the meaning of information, not just to store its content,” said Wilson, the Sherman Fairchild Professor of Neurobiology.
Memory Machinery
Tonegawa helped to usher in the physiological science of memory in 1992 when he and colleagues became the first to employ a genetic manipulation to demonstrate the pivotal role of a gene in memory formation. In mice, the scientists “knocked out,” or deleted, the gene alpha CaM kinase II, which enables neurons in the hippocampus to form and strengthen connections with other neurons. Mice missing the gene failed to learn key locations in a maze. At the time, knocking out genes was cutting edge, but also relatively crude. The CaMkII deletion occurred all over the mouse body. Neuroscientists gained the ability to target specific regions or cell types. In 1996, for the first time, a knockout was targeted to a specific cell type in a specific brain region: the NMDA receptor gene in hippocampus area CA1 pyramidal cells. In this way, Tonegawa and Wilson demonstrated that the gene is crucial for spatial memory coding and acquisition. In 2002, Tonegawa and Wilson further showed that a gene for the NMDA receptor was necessary in hippocampus area CA3 for mice to be able to retrieve spatial memories efficiently.
Neuroscientists have also become deft at editing genes and inserting new ones. In “optogenetics,” for example, genetic insertions enable neurons to become active or inactive in response to different colors of light.
“As work went on, the questions you could ask became more and more precise and specific,” Tonegawa said. “This opened up a new way to study memory in the brain.”
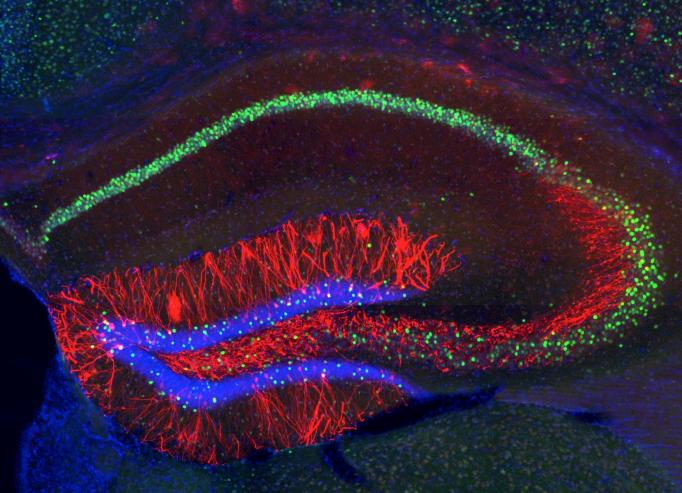
In 2012, Tonegawa’s lab was also the first to show that by labeling genes activated early in memory formation, they could pinpoint the ensemble of a hippocampal “engram,” the neurons that encoded a specific fear memory. How did they know? Because when they optogenetically activated the ensemble, the mouse froze as if recalling that fearful episode.
In subsequent studies, Tonegawa’s lab achieved other marvels of manipulation that demonstrate how memory is situated and structured in the brain. In one, the lab created a false memory by artificially reactivating the engram of a previously experienced place while giving the mouse a little shock. Mice then exhibited a fearful response in that place, even though they hadn’t actually experienced the shock there.
In a paper relevant to PTSD and anxiety, Tonegawa’s lab flipped the emotional association of memories. For instance, they gave a mouse a little shock in a context, initially conditioning fear. But then, when they reactivated the engram of that context while presenting a reward, the cells rewired their connection to the amygdala, a region that governs feelings, to produce a more positive association. The researchers also did the opposite, swapping an engram’s positive association for a fearful one.
In 2015, the lab highlighted the distinction between memory storage and recall. They showed that if they weakened the protein synthesis that enable engram cells to strengthen connections, or synapses, they could block a mouse’s ability to recall a memory. But if they artificially activated the engram they could still produce recall. The study demonstrated that synaptic strength may determine whether a memory is accessible, but the mere pattern of connections among the cells was enough to store it. In 2017, they reported that memory engram cell ensembles are simultaneously formed in the hippocampus and in the cortex immediately after an experience, challenging the widespread assumption that the episodic memory is first stored in the hippocampus and related brain regions, but the storage site shifts to the cortex only subsequently. This finding generated a new concept of “silent memory,” a memory state that cannot be retrieved efficiently by natural recall cues.
A simultaneous area of advancement has been observing and controlling “epigenetics,” the set of molecular factors that modify how genetic information is “expressed,” or implemented by cells. The lab of Picower Professor and Institute Director Li-Huei Tsai has used these advances to understand and improve memory storage and recall. Tsai, who studies neurodegenerative memory disorders such as Alzheimer’s disease (AD), has since helped to translate her insights into strategies to develop potential therapies.
In 2006, just as she was joining the Picower Institute, Tsai’s lab showed that placing AD model mice in a more mentally stimulating environment improved their flagging memory recall. The team sought the molecular mechanism. In mice that experienced enrichment, they observed, genes associated with synaptic function in learning and memory were in a more physically accessible state for expression, compared to the same DNA in unenriched animals. They then showed that by chemically inhibiting proteins responsible for locking down DNA, called HDACs, they could mimic enrichment’s benefits and improve memory recall.
“That was pretty neat because seemingly lost memories were not really lost,” Tsai said. “You could do something to get it back.”
In subsequent papers, the researchers advanced their understanding. In 2009, they determined that HDAC2 was the specific HDAC responsible and in 2012 they showed that Alzheimer’s pathology misregulates HDAC2, indicating one way the disease disrupts memory. In 2017 they found that they could inhibit HDAC2 specifically by inhibiting a co-factor called sp3. That finding pinpoints the specific nuclear complex that targets memory gene expression and helps to guide the pursuit of potential drugs to enhance memory while minimizing unintended effects on other HDACs.
Tsai’s investigation of gene expression in AD generated a surprising finding about the speed of memory formation. Her lab had observed that in many AD models, neural DNA showed the severe damage of double-strand breaks (DSBs) so the lab set out to induce DSBs in neurons to see how they affected gene expression. In 2015 Tsai and colleagues discovered that DSBs are necessary for the expression of genes that are expressed at the very beginning of memory formation. They essentially snap open DNA for rapid expression when activated by a memorable stimulus. In healthy mice, the breaks are repaired within hours. In AD, Tsai hypothesizes, the breaks are not repaired well, potentially leading to neural damage and death.
Memory Meaning
Like Tonegawa, Wilson has long been interested in pinpointing and studying specific memories in the hippocampus but does it differently. Beginning as a postdoc in Arizona in the early 1990s and continuing to this day, Wilson has pioneered methods of eavesdropping on and interpreting the electrical activity, or “firing,” of neurons to discern what that chatter of rising and dropping voltages encodes. Via these methods, Wilson’s lab can read out the location in a maze that a rat is remembering within mere millimeters. Last year, in fact, he and former postdocs showed they could read such thoughts almost in real-time.
“You can define memory based upon what’s left in the brain after you take away the world.”
Wilson’s goal has been to understand how memories become meaningful in intelligent behaviors such as pursuing reward.
He arrived at MIT and the CLM in 1994 just as he led a seminal paper showing that the same ensembles of hippocampal neurons in rats that would fire as the rat ran a maze, would also fire in the same patterns while the rat was asleep. Capturing this memory “replay” in action was a powerful demonstration that substantiated the hypothesis that memory is consolidated during sleep.
Sleep is an ideal condition for memory research, Wilson notes, not only because of its consolidation role but also because in the absence of stimulation or experience, whatever information the brain is processing can easily be recognized as memory.
“You can define memory based upon what’s left in the brain after you take away the world,” Wilson said.
Over time, Wilson’s work on memory replay has shown a great deal about what “processing” means. In a pair of papers in 2001 and 2002, for example, he showed that while memory replay in sleep’s REM phase occurs in long stretches almost like the timescale of real experience, replay in slow-wave sleep occurs at about 20 times speed and is broken up into short snippets.
In 2009 Wilson’s lab showed that rats replay memories of their journeys even while awake, during resting moments along the way. Given a moment to think, rats consider their environs, including remote parts of the maze. Notably, they even replay the maze in reverse, as if retracing their steps. Like in slow-wave sleep, the team found, memories were replayed rapidly and in small snippets, even as their order in time was preserved. Wilson hypothesizes that in these reflective moments the brain may be considering how fundamental units of experience can be regarded, understood and recombined for other uses.
“The idea that sleep carries some function is a mystery of memory,” Wilson said. “What goes on in these offline periods of introspective evaluation that contribute to higher level functions like creativity, insight and wisdom? How might that be accomplished? Decomposing and recomposing experience is attractive, and finding explicit neural correlates of this is supportive of this idea.”
Picower Professor Earl Miller joined the ranks of the CLM in 1995. He studies higher-level cognition where memory is put to use. Miller’s lab, for instance, examines how the brain sorts objects into categories, a feat that requires combining new sensory information with prior experience (i.e. the meaning of memory). Miller also studies the special system of working memory. The “sketchpad of consciousness,” working memory is how people intentionally select and manipulate information – both remembered and new – to accomplish tasks. It’s how, for instance, one formulates a plan for the day.
“When I first started the lab 25 years ago, the working memory task of holding something in mind for a while was the highest level cognitive phenomenon people were studying, but that’s all they were studying,” he said. “To me, the main story is how the animal knows to do what you told it to do. Nobody was studying that.”
Miller lab’s blends precise measurements of neural activity with measurements of broad scale brain oscillations, which are the rhythms that synchronize neurons to work together. The team focuses on areas of the cortex that process new sensory information as well as areas, like the prefrontal cortex, that engage in cognitive information processing.
Miller’s lab observed that sensory information is encoded among cortical neurons by high-frequency “gamma” rhythms, while the learned understanding of patterns that allow for categorizing a new object are encoded by lower-frequency “beta” rhythms. Meanwhile in 2014, a young Swedish neuroscientist named Mikael Lundqvist developed a computational model to hypothesize a way that working memory could hold multiple bits of information simultaneously in mind. He posited that oscillations occur in brief bursts, rather than persistently, with beta rhythms controlling gamma ones.
Walking to work one day, Miller realized that Lundqvist’s model squared well with his observations and predicted several working memory phenomena. Lundqvist joined the lab in 2015 and in a flurry of recent papers, Miller’s team has since experimentally confirmed many aspects of the model.
A grand culmination came in 2018 in the form of a new model of working memory. Governed by the memories and goals encoded in bursts of beta rhythms from neurons in deeper layers of the cortex, gamma waves encode new sensory information for consideration and juggle multiple (but limited) items of information in mind for processing. This hierarchical blending of bursts explains several phenomena that the orthodoxy of persistent activity does not, Miller said.
The model, dubbed “Working Memory 2.0,” could aid research on schizophrenia, where working memory is known to founder.
In the visual cortex (V1), Picower Professor Mark Bear’s lab has found the site of a fundamentally important type of memory that impacts behavior. “Visual recognition memory” helps us account for what we’ve already seen so that we are better able to focus on what’s new.
“We rely very much on our ability to detect novelty in a sea of familiarity,” he said. “To pick out the novel stimulus you have to have already formed a memory of what’s familiar.”
Notably, Bear said, this kind of memory appears to be disrupted in conditions like autism and schizophrenia.
Bear confesses that the discovery was somewhat of an accident. His then graduate student Nate Sawtell was trying to assay visual responses in V1 of awake mice when he encountered something confusing. Mice shown the same visual pattern every day produced a steadily increasing electrophysiological response in layer 4 of V1. When he tried a novel pattern, the response was back at the initial baseline level. Bear ultimately called this phenomenon “stimulus- selective response potentiation” or SRP.
In a paper in 2015, the lab showed what SRP meant. Mice are so excited by new visual patterns they will try to explore them. This behavioral response diminishes, or habituates, over days as the once novel pattern becomes familiar and SRP emerges in V1. The team went on to show that by blocking in V1 various synaptic processes and receptors – the same protocols that can be used to disrupt SRP – they could prevent habituation. In other words, the ability the mice developed to recognize and ignore familiar stimuli was based the ability of the visual cortex to form a visual recognition memory.
Whether it serves to help us recognize what’s new or as the basis of all that we know, memory’s importance as a subject of neuroscience research remains undeniable. The progress evident in 25 years of Picower Institute research has helped to ensure that even more progress will follow in the years ahead.
This story originally appeared in our Print Newsletter. Click here for a Free Subscription.