Even in the primary visual cortex, a brain region named for its specialized role in processing basic features of what the eyes see, not every neuron ends up answering the call to process properties of visual input. Maybe that’s because each neuron receives a wide variety of inputs via thousands of circuit connections, or “synapses,” and has to opt to respond to the visual information vs. something else. In a new study in mice, neuroscientists at The Picower Institute for Learning and Memory at MIT reveal how neurons that perform visual processing bring order to this input to get the job done.
Neuroscientists are keenly interested in what inputs, from among so many choices, will compel neurons to participate in the brain’s computations and functions, said senior author Mriganka Sur, Newton Professor of Neuroscience in The Picower Institute and MIT’s Department of Brain and Cognitive Sciences. Neurons ultimately participate in brain circuits by “firing” an electrical action potential.
“The configuration of inputs, the kind of organization, the assembly of neurons that modulate each other to generate an action potential is the essence of how brain circuits process information,” Sur said. “These (visual cortex) cells are a microcosm of this very profound and big picture of neuroscience.”
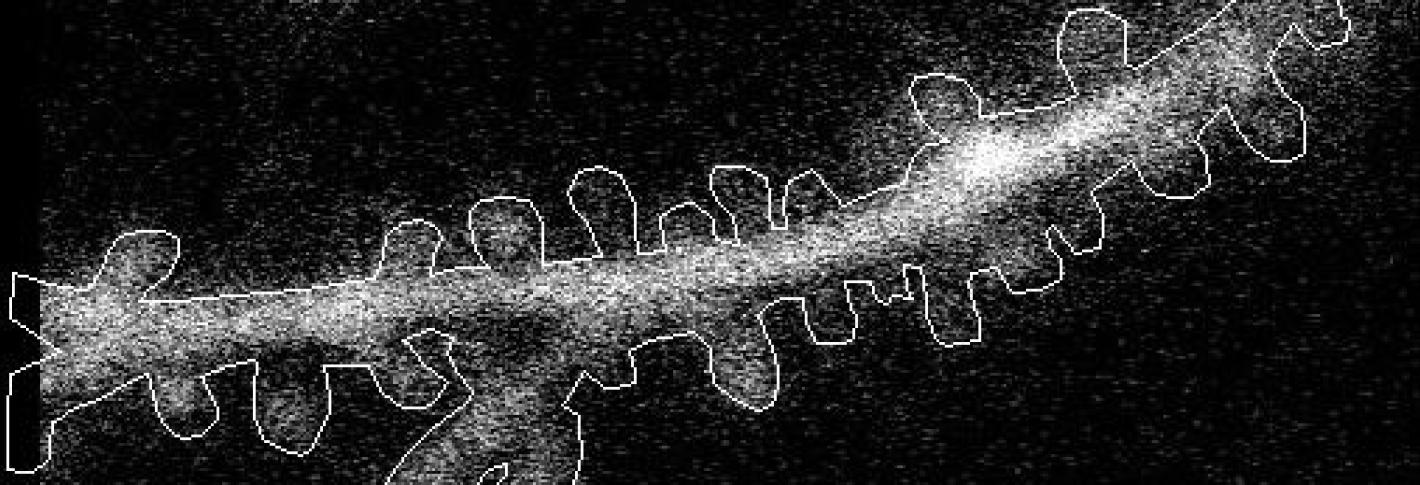
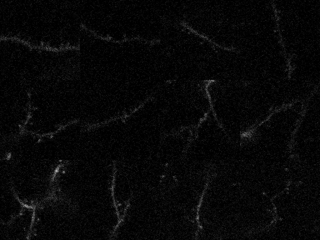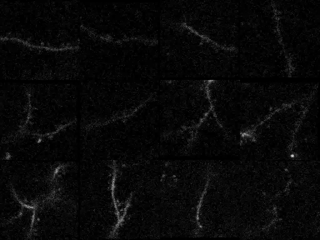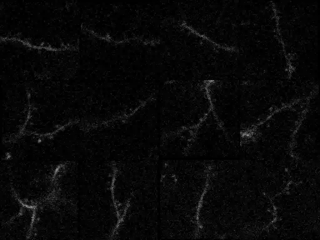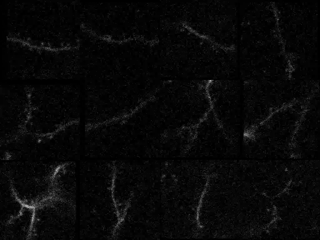
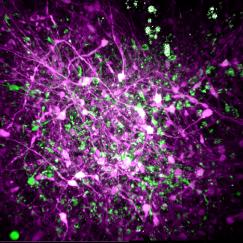
In the study in iScience, led by postdoc Kyle Jenks, the research team achieved their findings by meticulously imaging how not only neurons’ cell bodies but also how their individual synapses, formed on protrusions known as dendritic spines, responded as mice viewed moving images. They did this imaging for not only visually responsive neurons, but also for unresponsive neurons that nevertheless have visually responsive spines. That allowed them to analyze many key properties that might influence where a particular synapse forms, and how it influences responses at the cell body.
“This pulls together a lot of things that have been looked at in isolation and looks at them in one collective paper,” Jenks said. “We can compare how the neuron and the spines on that neuron respond to the same stimuli, and we can do this for both visually responsive and unresponsive neurons.”
Revealing rules
In visual cortex layer 2/3, Jenks and the team genetically engineered neurons such that their individual dendritic spines would glow when surges of calcium indicated increased activity by the synapses on the spines. The scientists did the same for the cell body, or “soma,” to keep track of how the cell responded and even signaled its overall responses back out to the synapses. This way, as the mice watched black and white gratings at varying angles drift by their eyes in different directions, the scientists could keep track of each spine’s and each cell’s overall response to that patterned visual input.
In all, they tracked 11 neurons that responded to the visual input and 11 others that seemingly ignored it. That enabled them to find several rules:
Distance from the soma matters: On cells that responded to visual input, the responses of individual spines were much more likely to correlate with the activity of the soma the closer the spine was to the soma. In the same vein, the soma’s signal back out to spines, which is believed to influence the spines’ alignment with the soma’s preferences, was more likely to be detectable closer to the soma than farther away.
Local clustering: On neurons that responded to visual input, spines formed distinct little enclaves of correlated responses with each other. Specifically, spines within 5 microns (5 millionths of a meter) acted in concert. But then, right outside that 5-micron boundary, spines were less likely than chance to join in that activity. Sur speculates that these isolated pockets of activity sharpened the response from each enclave.
“Apical” vs. “basal”: The neurons the team studied have two distinct kinds of dendrites. Apical dendrites, which are very long and protrude from the top, or “apex,” of the neuron, tend to get a wide variety of inputs from across the cortex. Basal dendrites, which are shorter and extend out from the bottom, typically get more raw visual input. While basal dendrites indeed received more visual input than apical dendrites overall, Jenks found that apical dendrites on visually responsive neurons had significantly more visually responsive spines than those on non-responsive neurons. And both types of dendrites equally obeyed the rules above about distance from the soma.
Orientation selectivity matters most: Jenks, Sur and the team used statistical modeling to determine which of many factors (the stimulus selectivity, reliability of the response, a spine’s distance from the soma, apical vs. basal, etc.) most explained how correlated a spine’s responsiveness was with that of the soma. By a wide margin, how selective a spine was to the orientation of its preferred grating was the most important single factor.
“Our results reveal that synaptic inputs to excitatory layer 2/3 neurons in mouse (visual cortex) are not randomly arranged, but organized and distributed in a manner that correlates with multiple factors including somatic responsiveness, somatic tuning, branch type, distance from the soma, local correlations, and stimulus selectivity,” the researchers wrote.
The team’s findings can help advance studies of vision in the brain in multiple ways, Jenks and Sur said. Certain genetic mutations that affect how neurons connect in circuits can affect visual cortex neurons and vision, Sur said. Documenting these rules provides researchers with a baseline to compare against when examining the effects of such mutations. Jenks added that the findings could inform efforts to model how neurons integrate synaptic inputs in their computations.
In addition to Sur and Jenks, the paper’s other authors are Gregg Heller, Katya Tsimring, Kendyll Martin, Asrah Rizvi, and Jacque Pak Kan Ip.
The National Institutes of Health, the Simons Foundation Autism Research Initiative and the Freedom Together Foundation provided support for the study.